Translate this page into:
Allergic reactions to radiocontrast media–A brief overview
*Corresponding author: Alpana Mohta, Department of Dermatology, Venerology and Leprosy, Sharda Multi Speciality Clinic, Bikaner, Rajasthan, India. dralpanamohta10@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Mohta A. Allergic reactions to radiocontrast media–A brief overview. Indian J Skin Allergy. 2024;3:128-30. doi: 10.25259/IJSA_4_2024
INTRODUCTION
The global utilization of iodinated radiocontrast media (RCM) surpasses 70 million annually, leading to frequent adverse responses.[1] The most commonly reported systemic adverse events following RCM administration are nephrotoxoicity and neurotoxicity.[2] Hypersensitivity reactions (HSRs) to RCM are categorized into immediate HR (IHR), and non-immediate HR (NIHR). IHR develops within 1–6 h of RCM administration, while NIHR typically has a delayed onset occurring at least 6 h after the administration. However, practically, the onset of NIHR is seen between 1 and 10 days.[3] A reported 0.73% overall prevalence of allergic reactions has been documented from RCM, with re-exposure being a significant risk factor.[4]
CLINICAL MANIFESTATIONS
The clinical significance of RCM-related hypersensitivity reactions (HSRs) ranges from mild urticaria and pruritus to anaphylaxis that is rarely fatal (estimated incidence of 1–2/100,000 procedures).[3,5]
Pruritus, urticaria, and/or angioedema are the most common presenting symptoms of RCM-induced IHR [Figure 1].[6] Manifestations of anaphylaxis, including wheezing, bronchospasm, hypotension, tachycardia, and small angioedema, are also reported.[6] Cardiac manifestations due to low blood flow include angina or acute coronary syndrome. Other manifestations include maculopapular exanthems, severe cutaneous adverse reactions, and iodine-induced sialadenitis.[7] NHIR generally manifests as non-severe maculopapular exanthema, urticaria, erythema multiforme, etc., [Figure 2]. However, in some cases, Stevens–Johnson syndrome, toxic epidermal necrolysis, acute generalized exanthematous pustulosis, and drug reactions with eosinophilia and systemic symptoms have also been reported. In patients allergic to iodine-based RCM, Gadolinium-Based Contrast Agents (GBCA) can be tried, as intravascular contrast in magnetic resonance imaging. However, these agents have also been associated with significant adverse effects, particularly nephrogenic systemic fibrosis (NSF), predominantly in patients with marked renal dysfunction. NSF primarily impacts the skin and occasionally internal organs in patients with reduced renal function. It presents as skin thickening and may cause organ dysfunction due to systemic fibrosis. Symptoms usually appear weeks to months after exposure to GBCA. Diagnosis relies on skin biopsy and clinical-histological analysis. Effective treatments are lacking, often leading to significant morbidity, despite potential symptom improvement post-acute kidney injury recovery or transplantation.[8]
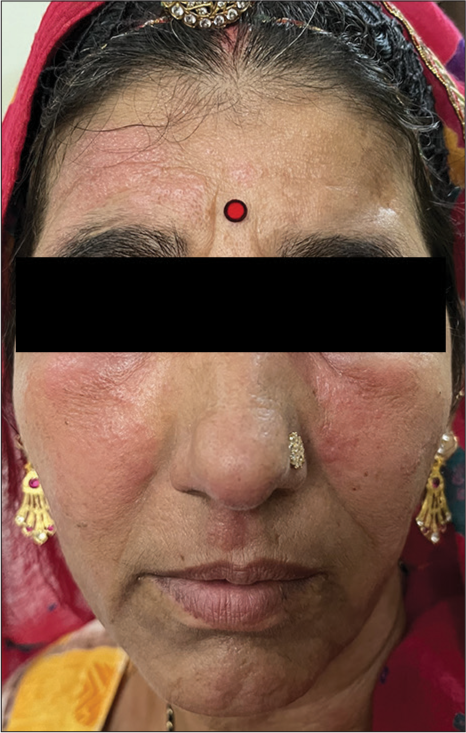
- Radiocontrast media induced immediate hypersensitivity reaction with severe angioedema.
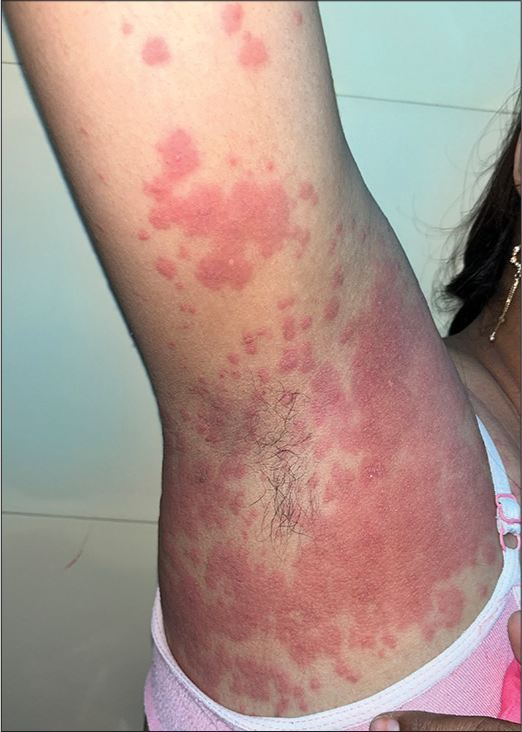
- Radiocontrast media induced delayed hypersensitivity reaction with localized erythema multiforme-like eruptions.
PATHOGENESIS
At present, available research indicates that the mechanism behind most cases of IHR is believed to be non-allergic. However, specific receptors Mas-related G protein-coupled receptor-X2 (MRGPRX2) were activated by RCMs iopamidol and iohexol in mouse models, suggesting a potential allergic mechanism. Positive skin tests, positive basophil activation tests, and raised levels of histamine and tryptase have been reported in cases exhibiting severe reactions. However, cases with mild-to-moderate IHR seldom report positive sensitization tests. Weak associations with genetic susceptibility genes and T cell-mediated hypersensitivity pathways are also reported.[9,10]
DIAGNOSTIC WORKUP
The recommended approach for testing patients who have experienced adverse reactions after receiving RCM depends on the manifestation of the symptoms. Patients with non-specific symptoms are unlikely to have severe reactions and may not require allergy testing. However, patients with typical symptoms of IHR or NIHR should undergo allergy testing. Skin tests are crucial for differentiation and safe alternative identification, with in vitro tests supporting the diagnosis.
Drug provocation tests (DPTs) are safe when conducted at experienced centers, but certain patient population require precautions.[3,9,11]
PROPHYLAXIS
Considering the propensity of some patients to exhibit reactions to multiple RCM, the challenge arises in identifying a non-reactive RCM without resorting to allergy diagnostics or drug provocation tests (DPT). Regrettably, current methodologies do not support this approach. Drug provocation test might not give reliable results simply because cross-reactivity exists not only among compounds from different chemical groups but also within the same group, albeit less commonly. Consequently, clinical decisions should prioritize the recognition of an individual’s likelihood of reacting to multiple contrast media (CMs) over the chemical structural similarities of the CMs.[12]
DESENSITIZATION IN ADULTS
Prophylactic desensitization in adults has been substantiated as effective, with a protocol encompassing diphenhydramine, prednisone, and ranitidine pretreatment. This is followed by a 13-step intravenous desensitization procedure. This regimen starts with a 10,000-fold diluted dose of RCM, incrementally doubled every 10 min.[13]
In hypersensitive patients, rapid drug desensitization involves escalating doses to reduce immunoglobulin E-mediated mast cell activation, aiming for the therapeutic or maximum tolerated dose. Historically used with taxanes and biologics like rituximab, it has a <10% chance of mild breakthrough reactions. Some protocols include premedication.
The role of premedication has been found to be beneficial in patients with prior immediate hypersensitivity reactions. It predominantly mitigates mild hypersensitivity responses, although it is not very effective against delayed or severe reactions. The standard adult premedication protocol includes 50 mg of oral prednisone administered at 13, 7, and 1 h before RCM exposure, and 1 mg/kg diphenhydramine administered either intramuscularly or orally 1 h prior.[14]
DESENSITIZATION IN CHILDREN
In pediatric patients, desensitization protocols have been adapted from adult models. The revised premedication regimen involves administering 0.5 mg/kg of methylprednisolone, orally or intravenously, at 13, 7, and 1 h before ICM administration. In addition, chlorphenamine is prescribed based on age: 1 mg (1–5 years), 2 mg (6–12 years), and 4 mg (>12 years), orally 1 h prior to RCM exposure.[14]
In severe anaphylaxis cases, RCM avoidance and allergy examination are recommended, with desensitization as an option in urgent situations. It is generally recommended that allergy testing be performed before RCM administration, and alternative RCMs should be administered without premedication in patients who are skin test-positive for the culprit RCM.[3,15,16]
MANAGEMENT
Management depends on the severity of the reaction. Switching the culprit RCM to a different agent is often been found to be more effective than premedication for mild reactions.[3,15,16]
Adverse reactions typically manifest as mild chemo-toxic responses, necessitating either mere observation or oral administration of H1-antihistamines. However, a small proportion of these reactions can escalate into critical anaphylactic events, necessitating heightened vigilance. Preparing emergency equipment and medication in advance is crucial.[17]
For mild hypersensitivity symptoms like itching, H1 antihistamines are generally effective. However, in the case of severe anaphylaxis, the administration of intramuscular epinephrine is vital, and many radiologists may not be aware of this necessity. The recommended initial dose is 0.01 mg/kg body weight, not exceeding 0.5 mg, of epinephrine at a 1:1000 concentration, to be administered intramuscularly in the thigh’s lateral aspect. Immediate assessment and management are crucial when symptoms affecting the airway, breathing, and circulation appear, with an emphasis on ensuring an adequate oxygen supply.[16,17]
Most NIHRs are mild-to-moderate and self-resolving, requiring minimal or no intervention. Severe cases such as Stevens–Johnson syndrome, toxic epidermal necrolysis, acute generalized exanthematous pustulosis, and drug reaction with eosinophilia and systemic symptoms necessitate symptomatic care, and require systemic corticosteroids, immunomodulators, intravenous immunoglobulins, and possible hospitalization.[16,17]
CONCLUSION
In patients testing negative on skin tests, a non-culprit RCM may be administered with or without premedication under emergency preparedness. Positive skin tests, especially in severe reactions, can help identify severe allergic reactions and guide future RCM selection. Avoiding the culprit and changing RCM based on allergy tests can enhance the safety of subsequent RCM exposures in patients with prior RCM hypersensitivity.
Ethical approval
The Institutional Review Board approval is not required.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The author confirms that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
Financial support and sponsorship
Nil.
References
- Side effects of radiographic contrast media: Pathogenesis, risk factors, and prevention. Biomed Res Int. 2014;2014:741018.
- [CrossRef] [Google Scholar]
- Allergy to radiocontrast dye. Immunol Allergy Clin North Am. 2022;42:391-401.
- [CrossRef] [PubMed] [Google Scholar]
- Hypersensitivity reactions to iodinated contrast media: A multicenter study of 196 081 patients. Radiology. 2019;293:117-24.
- [CrossRef] [PubMed] [Google Scholar]
- Fatal contrast medium-induced adverse response to iohexol in carotid artery angioplasty: A case report. Medicine (Baltimore). 2019;98:e16758.
- [CrossRef] [PubMed] [Google Scholar]
- Transient angioedema of the small bowel because of intravenous nonionic iodinated contrast media. J Pediatr. 2021;230:264-5.
- [CrossRef] [PubMed] [Google Scholar]
- Allergic-type reactions to radiographic contrast media. CMAJ. 2010;182:1328.
- [CrossRef] [PubMed] [Google Scholar]
- Radiographic contrast media and the kidney. Clin J Am Soc Nephrol. 2022;17:1234-42.
- [CrossRef] [PubMed] [Google Scholar]
- Immediate and delayed reactions to radiocontrast media: Is there an allergic mechanism? Immunol Allergy Clin North Am. 2009;29:453-68.
- [CrossRef] [PubMed] [Google Scholar]
- HLA-DRB1*15: 02 is associated with iodinated contrast media-related anaphylaxis. Invest Radiol. 2020;55:304-9.
- [CrossRef] [PubMed] [Google Scholar]
- Validation of the prescreening intradermal skin test for predicting hypersensitivity to iodinated contrast media: A prospective study with ICM challenge. J Allergy Clin Immunol Pract. 2020;8:267-72.
- [CrossRef] [PubMed] [Google Scholar]
- Cross-reactivity among iodinated contrast agents: Should we be concerned? Quant Imaging Med Surg. 2021;11:4028-41.
- [CrossRef] [PubMed] [Google Scholar]
- Rapid desensitization to overcome contrast allergy prior to urgent coronary angiography. Int Heart J. 2018;59:622-5.
- [CrossRef] [PubMed] [Google Scholar]
- Radiocontrast media hypersensitivity reactions in children. Medicina (Kaunas). 2022;58:517.
- [CrossRef] [PubMed] [Google Scholar]
- Relationship between lower dose and injection speed of iodinated contrast material for CT and acute hypersensitivity reactions: An observational study. Radiology. 2019;293:565-72.
- [CrossRef] [PubMed] [Google Scholar]
- Intradermal testing with radiocontrast media to prevent recurrent adverse reactions. AJR Am J Roentgenol. 2019;213:1187-93.
- [CrossRef] [PubMed] [Google Scholar]
- Hypersensitivity reactions to iodinated contrast media. Biomedicines. 2022;10:1036.
- [CrossRef] [PubMed] [Google Scholar]





